In the world of construction and civil engineering, the focus is often on the robustness of concrete presses or the accuracy of scales. However, there is one “invisible” ingredient that is involved in almost every operation: water.
But be careful, not all water is the same. Using tap water where distilled water is required by the standard can significantly skew your results.
1. What is distilled water (H₂O)?
Distilled water is water that has been purified through a process of distillation. Unlike drinking water, it has been stripped of its mineral salts (calcium, magnesium), organic impurities, and microorganisms.
Shutterstock
The difference with deionized water:
- Distilled water is obtained through evaporation and condensation. It is chemically and biologically pure (sterile).
- Demineralized water passes through ion exchange resins. It is chemically pure, but may still contain bacteria or pyrogens.
2. Why use it in your civil engineering tests?
The use of water containing ions (calcium, chlorine) may interact with chemical or physical samples.
For the Sand Equivalent Test
This is the most telling example. The cleaning solution must be diluted with high-purity water. The ions present in tap water can interfere with the flocculant, thereby altering the sedimentation rate of the clays.
Result: A distorted ES and sand potentially rejected incorrectly.
For determining Atterberg limits
When determining the liquidity limit of soil, the chemistry of the water used for humidification can alter the plasticity of clays. Distilled water ensures that only the nature of the soil is tested, not the composition of your local water supply.
For protecting your equipment
Using distilled water in your thermostatic baths or laboratory ovens extends their service life.
- Zero limescale: No scale buildup on heating elements.
- Zero corrosion: Less damage to stainless steel tanks.
3. Technical characteristics to watch out for
To be considered compliant in the laboratory, distilled water must meet certain criteria:
- Conductivity: It must be extremely low (generally
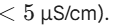
- Dry residue: Virtually none after evaporation.
- pH: Theoretically 7, it often drops to 5.5 because distilled water absorbs
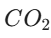 air, forming a very weak carbonic acid.
air, forming a very weak carbonic acid.
4. Conservation: The weak point
Distilled water is “hungry” for purity. As soon as it is exposed to air or stored in poor-quality containers, it becomes contaminated.
- CTRMAT advice: Store your water in clean, high-density polyethylene (HDPE) containers and keep them tightly closed.
Conclusion
Distilled water is not just a logistical detail; it is a guarantee of reliability and repeatability. By eliminating chemical variables related to water, you ensure the safety of your tests and the compliance of your construction sites.
Need to stock up your laboratory?
Find our distilled water at www.ctrmat.fr/shop/ctr102-eau-distillee-1-litre-1008.